
Malaria is a vector-borne infectious disease caused by protozoan parasites. It is widespread in tropical and subtropical regions, including parts of the Americas, Asia, and Africa. Each year, it causes disease in approximately 515 million people and kills between one and three million people, the majority of whom are young children in Sub-Saharan Africa.[1] Malaria is commonly associated with poverty, but is also a cause of poverty and a major hindrance to economic development.
Malaria is one of the most common infectious diseases and an enormous public health problem. The disease is caused by protozoan parasites of the genus Plasmodium. Only four types of the plasmodium parasite can infect humans; the most serious forms of the disease are caused by Plasmodium falciparum and Plasmodium vivax, but other related species (Plasmodium ovale, Plasmodium malariae) can also affect humans. This group of human-pathogenic Plasmodium species is usually referred to as malaria parasites.
Malaria parasites are transmitted by female Anopheles mosquitoes. The parasites multiply within red blood cells, causing symptoms that include symptoms of anemia (light headedness, shortness of breath, tachycardia etc.), as well as other general symptoms such as fever, chills, nausea, flu-like illness, and in severe cases, coma and death. Malaria transmission can be reduced by preventing mosquito bites with mosquito nets and insect repellents, or by mosquito control measures such as spraying insecticides inside houses and draining standing water where mosquitoes lay their eggs.
Although some are under development, no vaccine is currently available for malaria; preventative drugs must be taken continuously to reduce the risk of infection. These prophylactic drug treatments are often too expensive for most people living in endemic areas. Most adults from endemic areas have a degree of long-term recurrent infection and also of partial resistance; the resistance reduces with time and such adults may become susceptible to severe malaria if they have spent a significant amount of time in non-endemic areas. They are strongly recommended to take full precautions if they return to an endemic area. Malaria infections are treated through the use of antimalarial drugs, such as quinine or artemisinin derivatives, although drug resistance is increasingly common.
Symptoms
Symptoms of malaria include fever, shivering, arthralgia (joint pain), vomiting, anemia (caused by hemolysis), hemoglobinuria, and convulsions. There may be the feeling of tingling in the skin, particularly with malaria caused by P. falciparum. The classical symptom of malaria is cyclical occurrence of sudden coldness followed by rigor and then fever and sweating lasting four to six hours, occurring every two days in P. vivax and P. ovale infections, while every three for P. malariae.[30] P. falciparum can have recurrent fever every 36-48 hours or a less pronounced and almost continuous fever. For reasons that are poorly understood, but which may be related to high intracranial pressure, children with malaria frequently exhibit abnormal posturing, a sign indicating severe brain damage.[31] Malaria has been found to cause cognitive impairments, especially in children. It causes widespread anemia during a period of rapid brain development and also direct brain damage. This neurologic damage results from cerebral malaria to which children are more vulnerable.[32]
Severe malaria is almost exclusively caused by P. falciparum infection and usually arises 6-14 days after infection.[33] Consequences of severe malaria include coma and death if untreated—young children and pregnant women are especially vulnerable. Splenomegaly (enlarged spleen), severe headache, cerebral ischemia, hepatomegaly (enlarged liver), hypoglycemia, and hemoglobinuria with renal failure may occur. Renal failure may cause blackwater fever, where hemoglobin from lysed red blood cells leaks into the urine. Severe malaria can progress extremely rapidly and cause death within hours or days.[33] In the most severe cases of the disease fatality rates can exceed 20%, even with intensive care and treatment.[34] In endemic areas, treatment is often less satisfactory and the overall fatality rate for all cases of malaria can be as high as one in ten.[35] Over the longer term, developmental impairments have been documented in children who have suffered episodes of severe malaria.[36]
Chronic malaria is seen in both P. vivax and P. ovale, but not in P. falciparum. Here, the disease can relapse months or years after exposure, due to the presence of latent parasites in the liver. Describing a case of malaria as cured by observing the disappearance of parasites from the bloodstream can therefore be deceptive. The longest incubation period reported for a P. vivax infection is 30 years.[33] Approximately one in five of P. vivax malaria cases in temperate areas involve overwintering by hypnozoites (i.e., relapses begin the year after the mosquito bite).
Causes
Malaria parasites
Malaria is caused by protozoan parasites of the genus Plasmodium (phylum Apicomplexa). In humans malaria is caused by P. falciparum, P. malariae, P. ovale, and P. vivax. P. falciparum is the most common cause of infection, responsible for about 80 % of all malaria cases. However, P. falciparum is the most important cause of disease, and responsible for about 90% of deaths.[38] Parasitic Plasmodium species also infect birds, reptiles, monkeys, chimpanzees and rodents.[39] There have been documented human infections with several simian species of malaria, namely P. knowlesi, P. inui, P. cynomolgi,[40] P. simiovale, P. brazilianum, P. schwetzi and P. simium; however these are mostly of limited public health importance. Although avian malaria can kill chickens and turkeys, this disease does not cause serious economic losses to poultry farmers.[41] However, since being accidentally introduced by humans it has decimated the endemic birds of Hawaii, which evolved in its absence and lack any resistance to it.
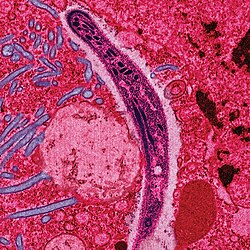
Diagnosis
The most economic, preferred, and reliable diagnosis of malaria is microscopic examination of blood films because each of the four major parasite species has distinguishing characteristics. Two sorts of blood film are traditionally used. Thin films are similar to usual blood films and allow species identification because the parasite's appearance is best preserved in this preparation. Thick films allow the microscopist to screen a larger volume of blood and are about eleven times more sensitive than the thin film, so picking up low levels of infection is easier on the thick film, but the appearance of the parasite is much more distorted and therefore distinguishing between the different species can be much more difficult. With the pros and cons of both thick and thin smears taken into consideration, it is imperative to utilize both smears while attempting to make a definitive diagnosis.
From the thick film, an experienced microscopist can detect parasite levels (or parasitemia) down to as low as 0.0000001% of red blood cells. Microscopic diagnosis can be difficult because the early trophozoites ("ring form") of all four species look identical and it is never possible to diagnose species on the basis of a single ring form; species identification is always based on several trophozoites. Please refer to the articles on each parasite for their microscopic appearances: P. falciparum, P. vivax, P. ovale, P. malariae.
In areas where microscopy is not available, or where laboratory staff are not experienced at malaria diagnosis, there are antigen detection tests that require only a drop of blood.[57] OptiMAL-IT® will reliably detect falciparum down to 0.01% parasitemia and non-falciparum down to 0.1%. Paracheck-Pf® will detect parasitemias down to 0.002% but will not distinguish between falciparum and non-falciparum malaria. Parasite nucleic acids are detected using polymerase chain reaction. This technique is more accurate than microscopy. However, it is expensive, and requires a specialized laboratory. Moreover, levels of parasitemia are not necessarily correlative with the progression of disease, particularly when the parasite is able to adhere to blood vessel walls. Therefore more sensitive, low-tech diagnosis tools need to be developed in order to detect low levels of parasitaemia in the field. Areas that cannot afford even simple laboratory diagnostic tests often use only a history of subjective fever as the indication to treat for malaria. Using Giemsa-stained blood smears from children in Malawi, one study showed that unnecessary treatment for malaria was significantly decreased when clinical predictors (rectal temperature, nailbed pallor, and splenomegaly) were used as treatment indications, rather than the current national policy of using only a history of subjective fevers (sensitivity increased from 21% to 41%).
Molecular methods are available in some clinical laboratories and rapid real-time assays (for example, QT-NASBA based on the polymerase chain reaction)are being developed with the hope of being able to deploy them in endemic areas.
Severe malaria is commonly misdiagnosed in Africa, leading to a failure to treat other life-threatening illnesses. In malaria-endemic areas, parasitemia does not ensure a diagnosis of severe malaria because parasitemia can be incidental to other concurrent disease. Recent investigations suggest that malarial retinopathy is better (collective sensitivity of 95% and specificity of 90%) than any other clinical or laboratory feature in distinguishing malarial from non-malarial coma.
Treatment
Active malaria infection with P. falciparum is a medical emergency requiring hospitalization. Infection with P. vivax, P. ovale or P. malariae can often be treated on an outpatient basis. Treatment of malaria involves supportive measures as well as specific antimalarial drugs. When properly treated, someone with malaria can expect a complete cure.
Antimalarial drugs
Further information: Antimalarial drugs
There are several families of drugs used to treat malaria. Chloroquine is very cheap and, until recently, was very effective, which made it the antimalarial drug of choice for many years in most parts of the world. However, resistance of Plasmodium falciparum to chloroquine has spread recently from Asia to Africa, making the drug ineffective against the most dangerous Plasmodium strain in many affected regions of the world. In those areas where chloroquine is still effective it remains the first choice. Unfortunately, chloroquine-resistance is associated with reduced sensitivity to other drugs such as quinine and amodiaquine.
There are several other substances which are used for treatment and, partially, for prevention (prophylaxis). Many drugs may be used for both purposes; larger doses are used to treat cases of malaria. Their deployment depends mainly on the frequency of resistant parasites in the area where the drug is used. One drug currently being investigated for possible use as an anti-malarial, especially for treatment of drug-resistant strains, is the beta blocker propranolol. Propranolol has been shown to block both Plasmodium's ability to enter red blood cell and establish an infection, as well as parasite replication. A December 2006 study by Northwestern University researchers suggested that propranolol may reduce the dosages required for existing drugs to be effective against P. falciparum by 5- to 10-fold, suggesting a role in combination therapies.
Currently available anti-malarial drugs include:
* Artemether-lumefantrine (Therapy only, commercial names Coartem® and Riamet®)
* Artesunate-amodiaquine (Therapy only)
* Artesunate-mefloquine (Therapy only)
* Artesunate-Sulfadoxine/pyrimethamine (Therapy only)
* Atovaquone-proguanil, trade name Malarone (Therapy and prophylaxis)
* Quinine (Therapy only)
* Chloroquine (Therapy and prophylaxis; usefulness now reduced due to resistance)
* Cotrifazid (Therapy and prophylaxis)
* Doxycycline (Therapy and prophylaxis)
* Mefloquine, trade name Lariam (Therapy and prophylaxis)
* Primaquine (Therapy in P. vivax and P. ovale only; not for prophylaxis)
* Proguanil (Prophylaxis only)
* Sulfadoxine-pyrimethamine (Therapy; prophylaxis for semi-immune pregnant women in endemic countries as "Intermittent Preventive Treatment" - IPT)
* Hydroxychloroquine, trade name Plaquenil (Therapy and prophylaxis)
The development of drugs was facilitated when Plasmodium falciparum was successfully cultured.[65] This allowed in vitro testing of new drug candidates.
Extracts of the plant Artemisia annua, containing the compound artemisinin or semi-synthetic derivatives (a substance unrelated to quinine), offer over 90% efficacy rates, but their supply is not meeting demand.[66] One study in Rwanda showed that children with uncomplicated P. falciparum malaria demonstrated fewer clinical and parasitological failures on post-treatment day 28 when amodiaquine was combined with artesunate, rather than administered alone (OR = 0.34). However, increased resistance to amodiaquine during this study period was also noted.[67] Since 2001 the World Health Organization has recommended using artemisinin-based combination therapy (ACT) as first-line treatment for uncomplicated malaria in areas experiencing resistance to older medications. The most recent WHO treatment guidelines for malaria recommend four different ACTs. While numerous countries, including most African nations, have adopted the change in their official malaria treatment policies, cost remains a major barrier to ACT implementation. Because ACTs cost up to twenty times as much as older medications, they remain unaffordable in many malaria-endemic countries. The molecular target of artemisinin is controversial, although recent studies suggest that SERCA, a calcium pump in the endoplasmic reticulum may be associated with artemisinin resistance.[68] Malaria parasites can develop resistance to artemisinin and resistance can be produced by mutation of SERCA.[69] However, other studies suggest the mitochondrion is the major target for artemisinin and its analogs.
In February 2002, the journal Science and other press outlets[71] announced progress on a new treatment for infected individuals. A team of French and South African researchers had identified a new drug they were calling "G25".[72] It cured malaria in test primates by blocking the ability of the parasite to copy itself within the red blood cells of its victims. In 2005 the same team of researchers published their research on achieving an oral form, which they refer to as "TE3" or "te3".[73] As of early 2006, there is no information in the mainstream press as to when this family of drugs will become commercially available.
In 1996, Professor Geoff McFadden stumbled upon the work of British biologist Ian Wilson, who had discovered that the plasmodia responsible for causing malaria retained parts of chloroplasts,[74] an organelle usually found in plants, complete with their own functioning genomes. This led Professor McFadden to the realisation that any number of herbicides may in fact be successful in the fight against malaria, and so he set about trialing large numbers of them, and enjoyed a 75% success rate.
These "apicoplasts" are thought to have originated through the endosymbiosis of algae[75] and play a crucial role in fatty acid bio-synthesis in plasmodia.[76] To date, 466 proteins have been found to be produced by apicoplasts[77] and these are now being looked at as possible targets for novel anti-malarial drugs.
Although effective anti-malarial drugs are on the market, the disease remains a threat to people living in endemic areas who have no proper and prompt access to effective drugs. Access to pharmacies and health facilities, as well as drug costs, are major obstacles. Médecins Sans Frontières estimates that the cost of treating a malaria-infected person in an endemic country was between US$0.25 and $2.40 per dose in 2002.
Counterfeit drugs
Sophisticated counterfeits have been found in Thailand, Vietnam, Cambodia[79] and China,[80] and are an important cause of avoidable death in these countries.[81] There is no reliable way for doctors or lay people to detect counterfeit drugs without help from a laboratory. Companies are attempting to combat the persistence of counterfeit drugs by using new technology to provide security from source to distribution.
Prevention and disease control
Methods used to prevent the spread of disease, or to protect individuals in areas where malaria is endemic, include prophylactic drugs, mosquito eradication, and the prevention of mosquito bites. There is currently no vaccine that will prevent malaria, but this is an active field of research.
Many researchers argue that prevention of malaria may be more cost-effective than treatment of the disease in the long run, but the capital costs required are out of reach of many of the world's poorest people. Economic adviser Jeffrey Sachs estimates that malaria can be controlled for US$3 billion in aid per year. It has been argued that, in order to meet the Millennium Development Goals, money should be redirected from HIV/AIDS treatment to malaria prevention, which for the same amount of money would provide greater benefit to African economies.[82]
Efforts to eradicate malaria by eliminating mosquitoes have been successful in some areas. Malaria was once common in the United States and southern Europe, but the draining of wetland breeding grounds and better sanitation, in conjunction with the monitoring and treatment of infected humans, eliminated it from affluent regions. In 2002, there were 1,059 cases of malaria reported in the US, including eight deaths. In five of those cases, the disease was contracted in the United States. Malaria was eliminated from the northern parts of the USA in the early twentieth century, and the use of the pesticide DDT eliminated it from the South by 1951. In the 1950s and 1960s, there was a major public health effort to eradicate malaria worldwide by selectively targeting mosquitoes in areas where malaria was rampant.[83] However, these efforts have so far failed to eradicate malaria in many parts of the developing world - the problem is most prevalent in Africa.
Brazil, Eritrea, India, and Vietnam have, unlike many other developing nations, successfully reduced the malaria burden. Common success factors included conducive country conditions, a targeted technical approach using a package of effective tools, data-driven decision-making, active leadership at all levels of government, involvement of communities, decentralized implementation and control of finances, skilled technical and managerial capacity at national and sub-national levels, hands-on technical and programmatic support from partner agencies, and sufficient and flexible financing.[84]
The Malaria Control Project is currently using downtime computing power donated by individual volunteers around the world (see Volunteer computing and BOINC) to simulate models of the health effects and transmission dynamics in order to find the best method or combination of methods for malaria control. This modeling is extremely computer intensive due to the simulations of large human populations with a vast range of parameters related to biological and social factors that influence the spread of the disease. It is expected to take a few months using volunteered computing power compared to the 40 years it would have taken with the current resources available to the scientists who developed the program.
No comments:
Post a Comment